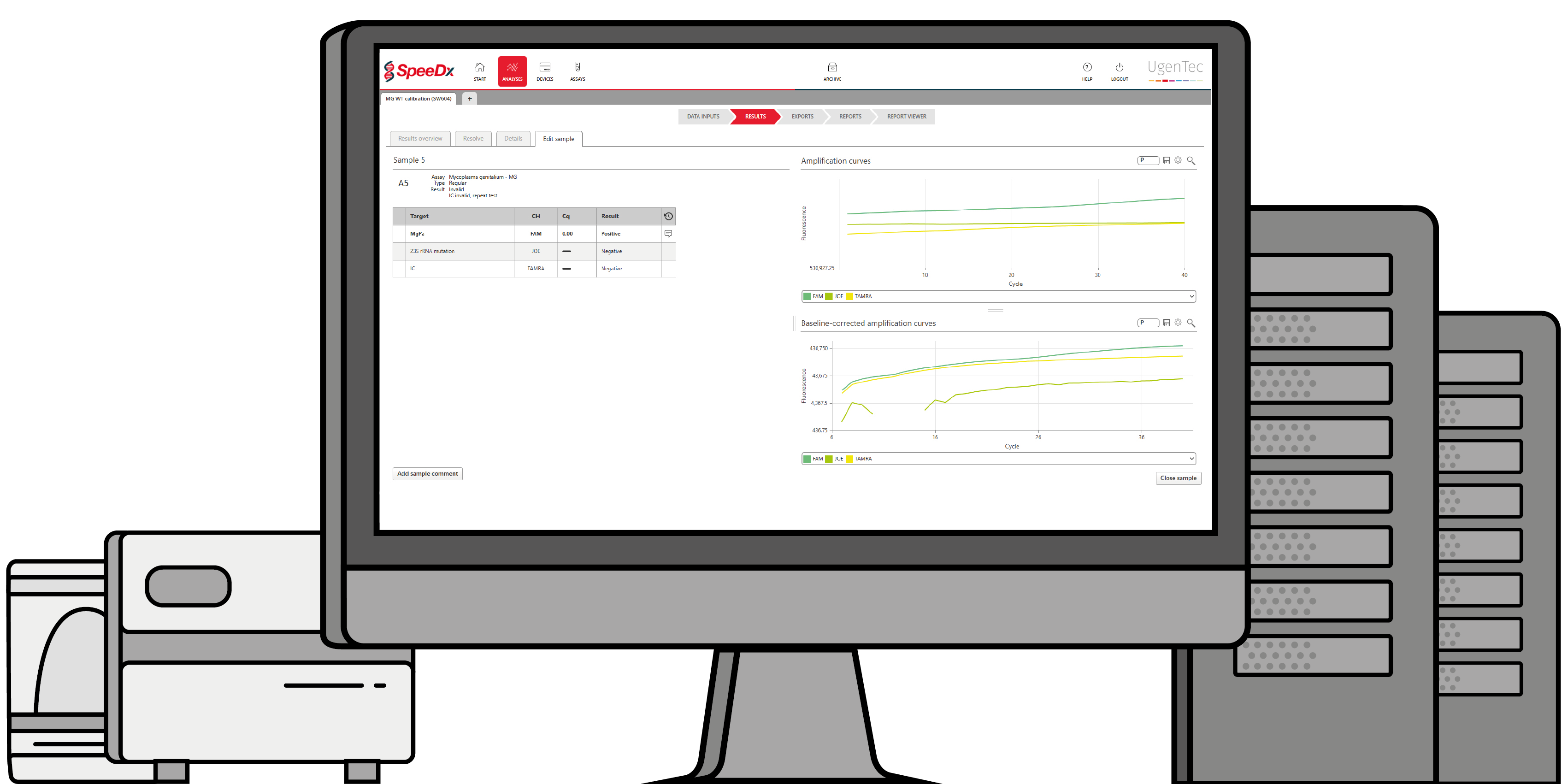
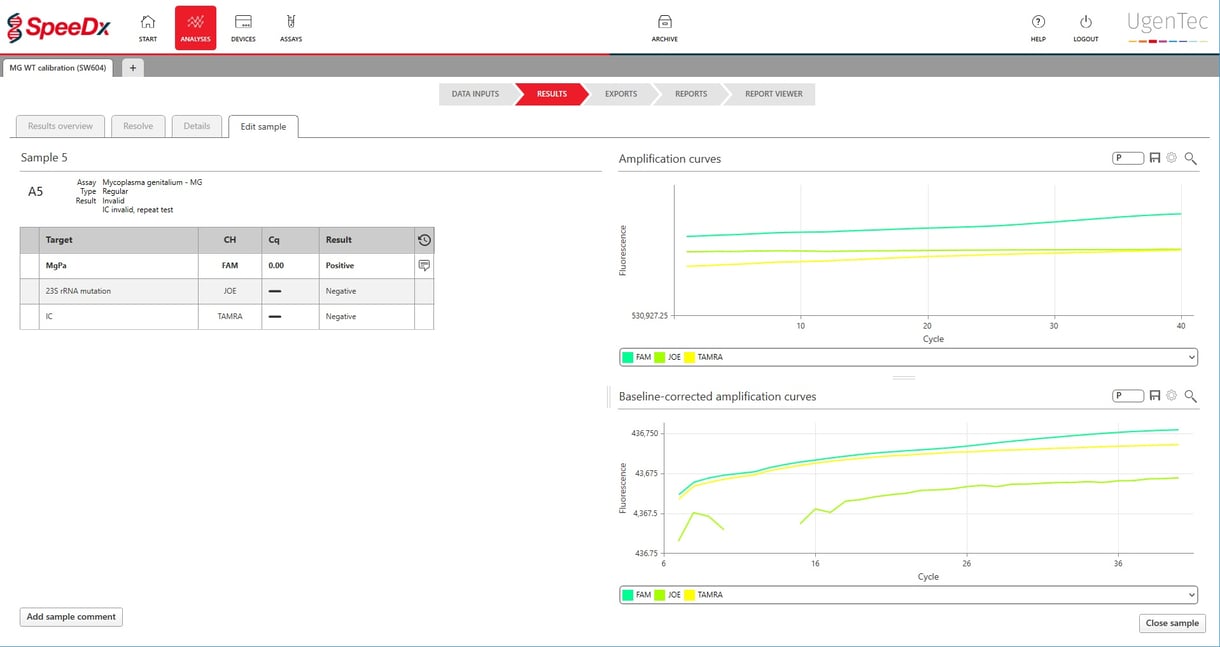
UgenTec NV, the European bio-informatics company that develops artificially intelligent PCR interpretation software, and SpeeDx Pty. Ltd., a developer of innovative molecular diagnostic solutions, today announced the launch of a smart interpretation module for the ResistancePlus™ MG test.
This diagnostic test detects both the sexually transmitted infection (STI) Mycoplasma genitalium and mutations in the 23S rRNA gene of the bacteria that have been shown to confer resistance to azithromycin, a commonly prescribed macrolide-based antibiotic. UgenTec’s solution, called FastFinder, ensures customers can automatically interpret the results and provides automation for incorporation to many laboratory information systems.
“Combining our high performance and reliable assays with UgenTec's software is unique in today's diagnostic landscape," says Colin Denver, CEO of SpeeDx. “We can now provide a validated solution for multiple qPCR instruments to help speed up the delivery of results during routine diagnostic use.” In the near future, SpeeDx intends to expand the interpretation module towards their pipeline of PlexPCRTM HSV-1&2, VZV, Syphilis and PlexPCRTM RespiVirus.
“UgenTec is looking forward to providing the customers of SpeeDx with an artificially intelligent assistant. It will provide them with powerful machine learning algorithms," said Wouter Uten, CEO of UgenTec. “this, combined with automated decision trees, ensures that end-users can maximize measurement certainty and decrease time to result.".
About SpeedX
SpeeDx is a privately owned company founded in 2009. Based in Sydney with a subsidiary office in London, SpeeDx specialises in innovative multiplex real-time polymerase chain reaction (qPCR) solutions for clinical diagnostics. With CE-IVD multiplex qPCR kits for detection of infectious disease pathogens and antimicrobial resistance markers, SpeeDx has a portfolio of market leading detection and priming technologies to enable new healthcare paradigms based on improved delivery and reduced costs. SpeeDx has a proven track record of scientific discovery and strives to provide cutting edge clinically relevant tools for the clinical diagnostic market.