For labs, task forces, and MDx providers
With lab professionals under strain to deploy reliable COVID tests, software is crucial to scale up test volumes.
As many county, state and national COVID testing initiatives in the US, Canada and Europe are coming online, task forces are marshaling existing infrastructure to hyper-scale PCR testing labs.
They are choosing FastFinder for sample-to-answer automation.
Supply chain issues are hurting closed sample-to-answer systems. COVID testing initiatives now rely on using available PCR systems.
Guarantee consistency of POS/NEG results across instrument types.
UgenTec FastFinder is the platform of choice for a growing number of large-scale COVID hubs to build sample-to-answer systems.
Ensure proper operation with any LIMS, chemistry, robotics, cycler, and COVID protocol.
With volumes scaling to 10K or 100K assays per day, there's a central challenge: operational efficiency. Standardized, at scale, and automated.
Deploy testing pipelines that very rapidly provide POS/NEG/retest answers.
UgenTec supports the diagnostic community in managing the Coronavirus crisis. Automate the CDC, Pasteur, Charité and other protocols. On any cycler. For any sample volume.
Whether you're implementing the CDC, Charite, Pasteur or other protocols - or are bridging to a custom implementation - we've got your back with protocol automation and data analysis from sample to answer.
You may run on different cyclers. Or 384 instead of 96 wells. Or your chemistry may vary. Rest assured - with FastFinder, you can build a sample-to-answer system regardless of the PCR instrumentation, chemistry or data analysis protocol.
Scale your sample volumes to unprecedented levels. Without adding hands-on time. The vast majority of samples are processed from raw data to POS/NEG result, and the exceptions are flagged fro review.
With FastFinder, molecular biologists can review exceptions and sign-off results from anywhere.
With smart connectivity and hosted infrastructure, FastFinder is ideal for multi-site and multi-lab setups. With the added benefit of cross-site dashboards on all key metrics, including positive rates, QC, instrument specific performance, and more.
FastFinder analyzes a single PCR plate in under 30 seconds. No more manual review and data entry, at scale, this means hours of time saved per technician.
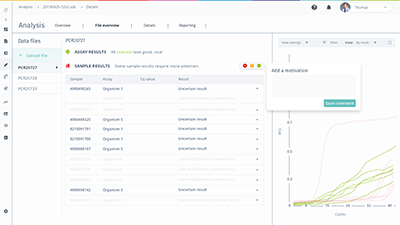
FastFinder Analysis takes any qPCR test in your menu and bolts on data analysis and interpretation support. For any qPCR device, for any protocol.
Fast, reliable, reproducible results: smart data analysis automation cutting down manual review and data entry.
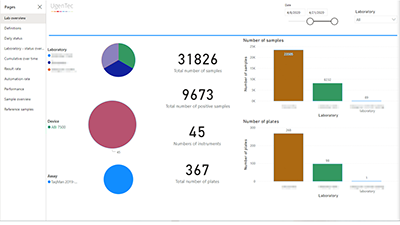
FastFinder Insights gives labs, MDx companies & testing task forces deep insights in cross-site operational performance, statistics and QC metrics.
Dashboards, reports and visualizations provide comprehensive intel across your entire assay menu, test results and statuses, instruments, operators and sites.
CDC: Instructions for running PCR detection assays in your laboratory
News: FDA issues emergency use authorization for CDC method.
CDC: RT-PCR panel, primers and probes for testing Coronavirus
Charité protocol: with Fastfinder, automate any of the published COVID protocols.